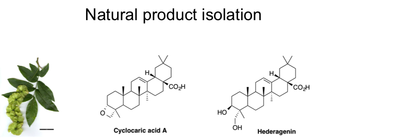
One project involves the isolation and identification of active components of extracts obtained from botanical samples used in Traditional Chinese Medicine (TCM). Through our affiliation with the Tennessee Center for Botanical Medicine Research, we have access to extracts used in TCM. Fractionation of these crude extracts has led to pure compounds in some cases, with bioactivity in diverse areas such as antiviral, antitrypanosomal and antiinflammatory as well as antitumor. One example is the isolation of hederagenin from Cyclocarya paliurus, which led to a correction of the structure of cyclocaric acid A. This oxetane had been reported originally as a component of C. paliurus, however we found that the plant isolate was actually hederagenin, a common oleanane with known antitumor activity.
Independent synthesis of cyclocaric acid A was used for confirmation. Collaboration with members of the Biology department is ongoing and we are currently investigating a number of extracts which are at varying stages of purification.
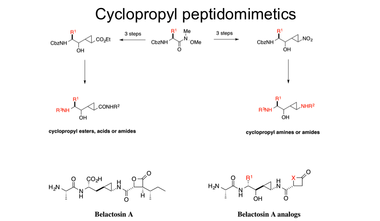
One area interest is in the synthesis of potentially bioactive compounds. Three synthetic projects are currently underway. The major project is focused on the design and synthesis of cyclopropyl-containing peptidomimetics as potential enzyme inhibitors. An efficient (four step) approach to a core structure has been developed, using protected amino acids as starting materials. The key steps involve either a sulfur ylide cyclopropantion or a nitrocyclopropanation. Differentiation at three variable points leads to compounds with potential as HCV NS3 protease or BACE inhibitors. We are also particularly interested in using the cyclopropanation methods for the synthesis of analogs of the proteasome inhibitor belactosin A.
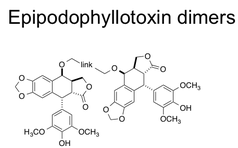
A second synthetic project involves the synthesis of dimers of 4’-demethylepipodophyllotoxin. A series of ether-linked dimers and their corresponding monomers were prepared and analyzed for DNA cleavage ability as well as tumor cytotoxicity. The initial series of dimers exhibited higher levels of double-strand cleavage as well as enhanced cytotoxicity over the monomers, lending credence to the two-drug model of interaction with topoisomerase II
